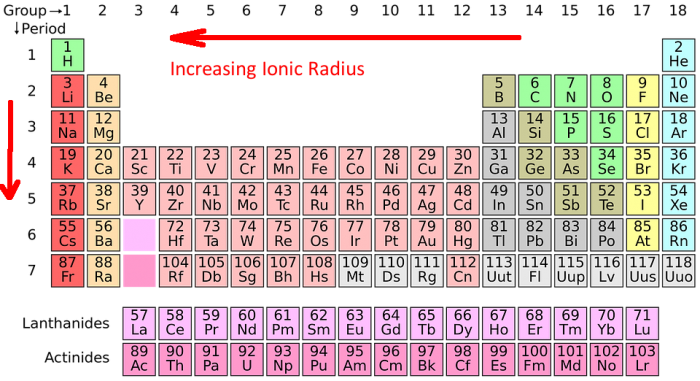
The ionic radius of metals (including transition metals) tends to decrease with increasing atomic number as they lose electrons, or as they lose their outer shell electron, whereas the ionic radius of non metals increases with increasing atomic number as they gain electrons, but only by a very small amount, so it can be considered negligible.(Although group 12 metals do not have partially filled d shells, their chemistry is comparable to that of the preceding groups in many aspects, therefore they are included in our explanation.) In contrast to the s and p block elements, the transition metals share considerable horizontal as well as vertical chemical similarities. Transition metals, which are found in groups 3-12 of the periodic chart, have partially filled d subshells in their free elements or cations. 
Ionic Radius trends in transition elements An anion, on the other hand, is larger than the parent atom because the addition of one or more electrons leads to increased electron repulsion and a lower nuclear charge. Because it has fewer electrons than its parent atom, the cation is usually smaller in size, while having the same nuclear charge. The distance between the electron in an ion’s outermost shell and the nucleus’ core is known as the ionic radius. Distances between cations and anions in ionic crystals can be used to estimate the ionic radius.In the same way, obtaining an electron causes the anion to form. As a result of the removal/loss of electrons, a cation is formed. An ion is formed when an atom gains or loses electrons. An ion is a net electrically charged atom, molecule, or particle. The ionic radius is defined as the radial distance at which an ion’s nucleus exerts an effect on its electron cloud. An ion is formed when an atom gets or loses an electron from its outer orbit in order to achieve a stable electrical state.Those in groups 7 and 10 have atomic radii that are somewhat similar, but those in groups 11 and 12 have bigger radii. Because of the poor shielding provided by the tiny amount of d- electrons, the transition elements’ atomic and ionic radii fall from group 3 to9. Transition elements are defined by IUPAC as elements with a partially filled d subshell or elements that have the ability to generate stable cations with an incompletely filled d orbital. Transition elements are elements with partially filled d orbitals (sometimes known as transition metals).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
